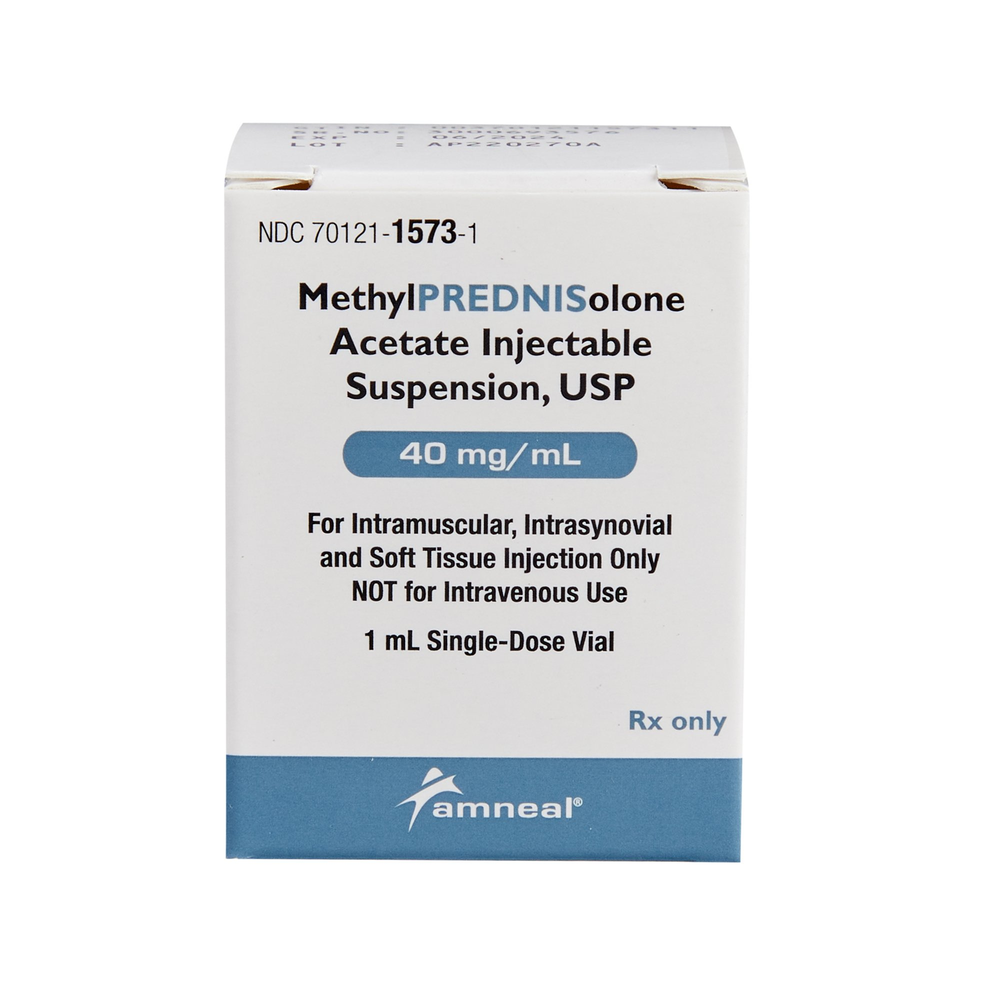
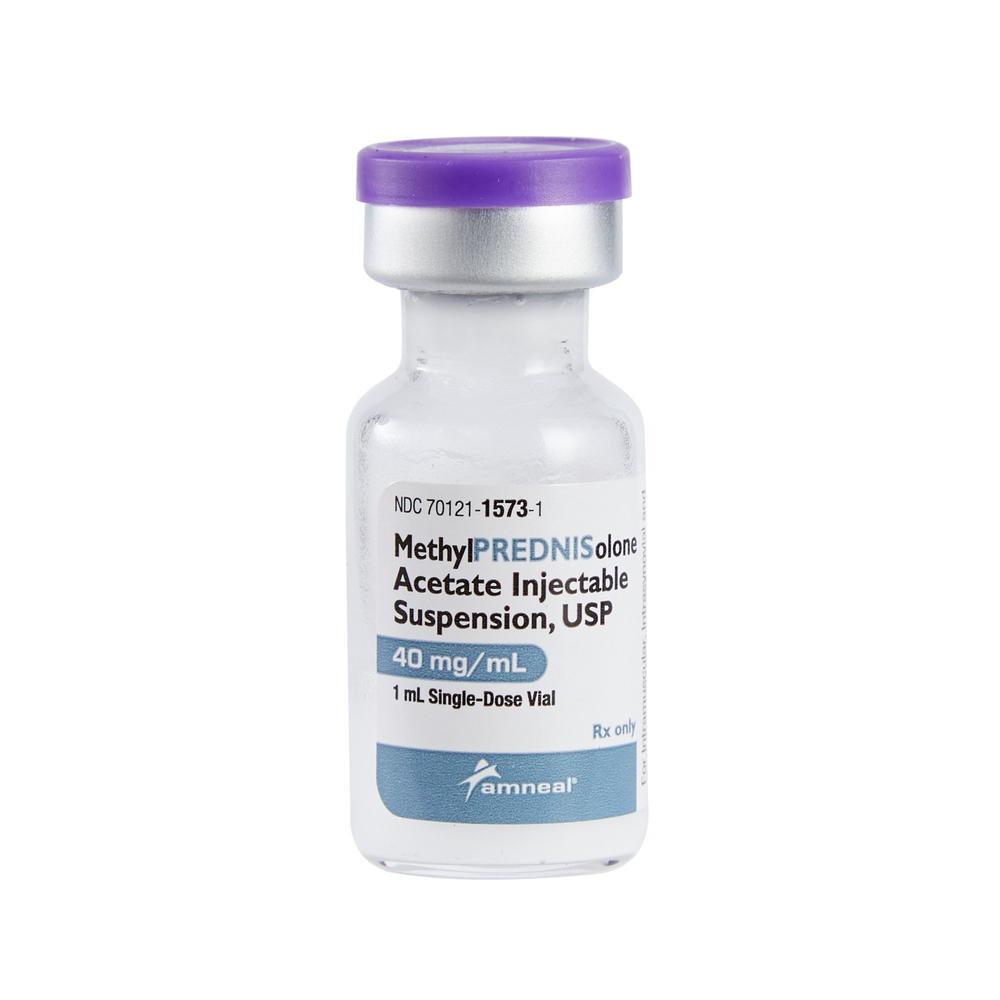
Product Specifications
| Specification | Details |
| Manufacturer / Brand | Amneal Pharmaceuticals, LLC |
| Manufacturer # / SKU | 70121157301 |
| NDC Number | 70121-1573-01 (Vial) / 70121-1573-1 (Box) |
| Active Ingredient | Methylprednisolone Acetate |
| Strength | 40 mg / mL |
| Volume | 1 mL |
| Container Type | Single-Dose Vial (SDV) |
| Formulation | Sterile Aqueous Suspension |
| Application | IM / Intra-synovial / Soft Tissue Injection |
| Storage | 20°C to 25°C (68°F to 77°F); Do not freeze |
Key Features
-
Sustained Release: The acetate ester provides a slower rate of absorption, allowing for prolonged therapeutic activity at the site of injection.
-
Versatile Administration: Indicated for various routes including intramuscular (systemic) and intra-articular (localized joint) injections.
-
Single-Dose Safety: Packaged in a 1 mL single-dose vial to ensure sterility and minimize the risk of cross-contamination.
-
Potent Anti-Inflammatory: Effectively suppresses symptoms of intense inflammation, particularly in rheumatologic and dermatologic disorders.
-
High Tactile Control: Formulated as a smooth suspension for consistent flow through fine-gauge needles.
Clinical Applications
-
Rheumatoid Arthritis: Short-term adjunctive therapy for acute episodes.
-
Osteoarthritis: Intra-articular injection for localized relief of pain and inflammation.
-
Severe Allergic Reactions: Systemic control of conditions like bronchial asthma or contact dermatitis.
-
Dermatologic Conditions: Used for intralesional treatment of localized hypertrophic, inflammatory, or lichenified lesions.
⚠️ Usage Note: NOT FOR INTRAVENOUS (IV) USE. This is an aqueous suspension and must only be used for IM, intra-articular, or soft tissue injection.
⚠️ Safety Alert: Use strict aseptic technique. Do not use if the suspension has clumped or changed color.
⚠️ Contraindication: Do not administer to patients with systemic fungal infections or those with hypersensitivity to methylprednisolone.



Reviews
There are no reviews yet.