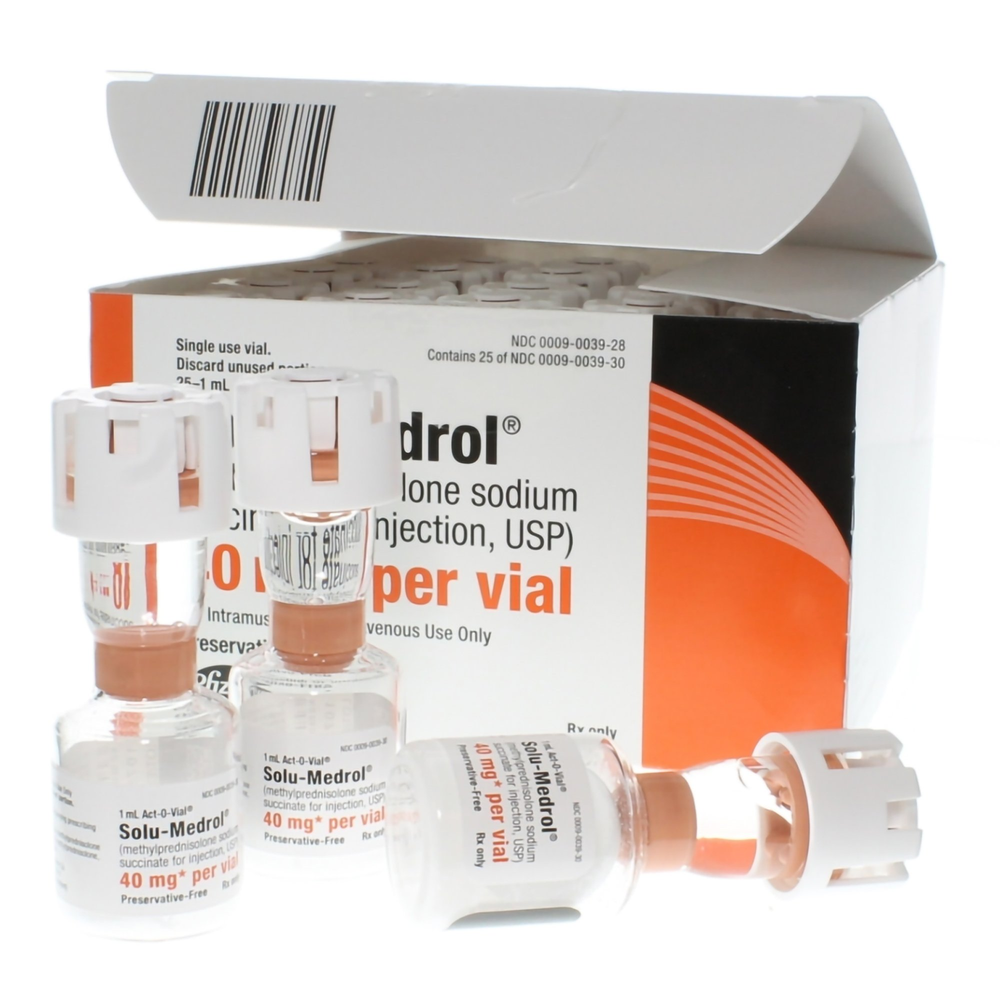
Product Specifications
| Specification | Details |
| Manufacturer / Brand | Pfizer Inc / Solu-Medrol® |
| Manufacturer # / SKU | 00009003928 |
| NDC Number | 00009-0039-28 |
| Active Ingredient | Methylprednisolone Sodium Succinate |
| Strength | 40 mg / 1 mL |
| Formulation | Preservative-Free Sterile Powder |
| Container Type | Act-O-Vial® (Dual-Chamber Vial) |
| Route of Admin | IV (Intravenous) or IM (Intramuscular) |
| Packaging | 25 Vials per Carton |
| Latex Content | Not made with natural rubber latex |
Key Features
-
Instant Reconstitution: The Act-O-Vial® system allows for immediate mixing without the need for additional syringes or diluent vials, reducing the risk of contamination and dosage errors.
-
Rapid-Onset Relief: Provides swift anti-inflammatory and immunosuppressive effects, essential for treating acute allergic reactions or inflammatory flares.
-
Preservative-Free: Ideal for sensitive patients; however, it should be used immediately or within 48 hours of reconstitution if refrigerated.
-
Versatile Administration: Suitable for direct IV injection, IV infusion (when further diluted), or deep IM injection.
-
Precise Small-Volume Dosing: The 40 mg/1 mL concentration is perfect for pediatric protocols and targeted adult dosing.
Clinical Applications
-
Allergic States: Management of severe asthma, anaphylaxis, and drug hypersensitivity reactions.
-
Dermatologic Diseases: Treatment of Stevens-Johnson syndrome and severe exfoliative dermatitis.
-
Endocrine Disorders: Secondary adrenocortical insufficiency and congenital adrenal hyperplasia.
-
Rheumatic Disorders: Adjunctive therapy for acute episodes of rheumatoid arthritis or gouty arthritis.
⚠️ Black Box & Safety Warnings: Not for Intrathecal Use. Administration by this route has been associated with severe medical complications.
⚠️ Usage Note: Press down on the plastic activator to force the diluent into the lower chamber. Gently agitate to ensure the solution is clear before withdrawing into a syringe.
⚠️ Contraindications: Contraindicated in patients with systemic fungal infections or known hypersensitivity to any of the ingredients.