Product Specifications
| Specification | Details |
| Manufacturer / Brand | Amneal Pharmaceuticals, LLC |
| Manufacturer # / SKU | 70121161001 |
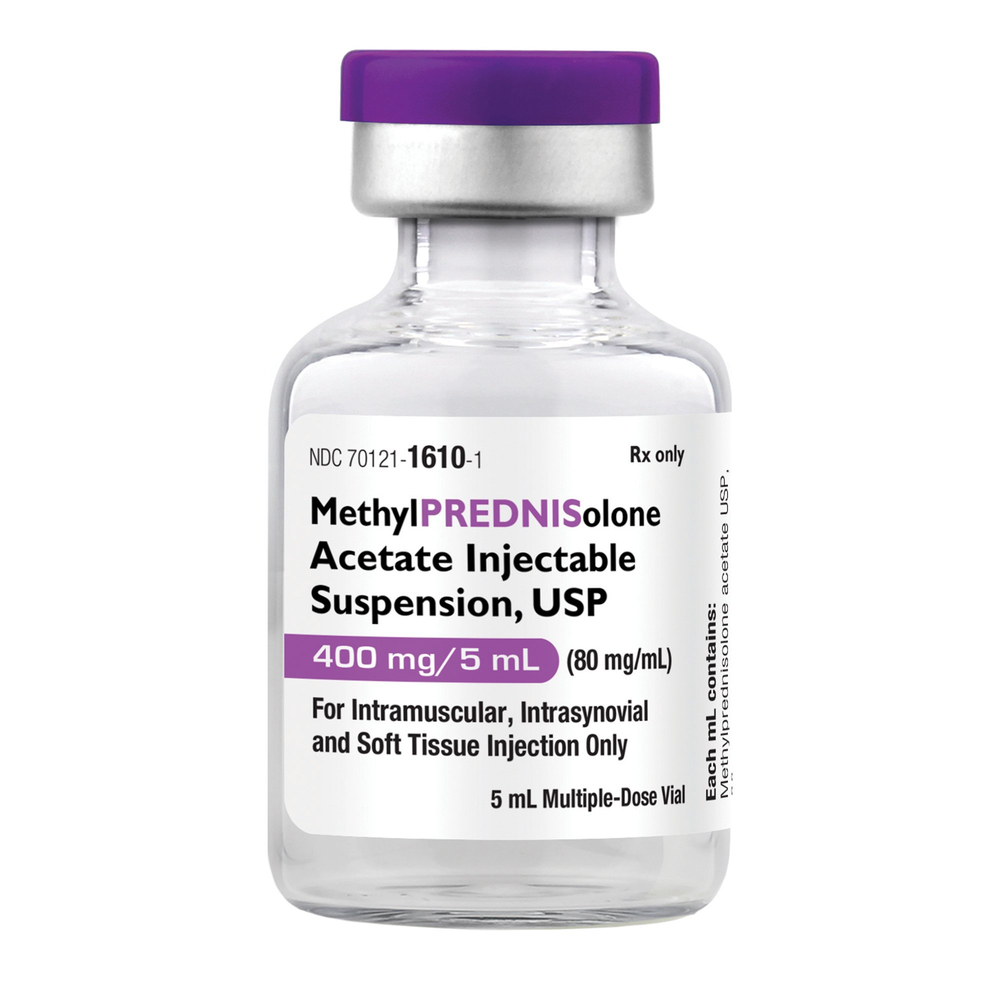
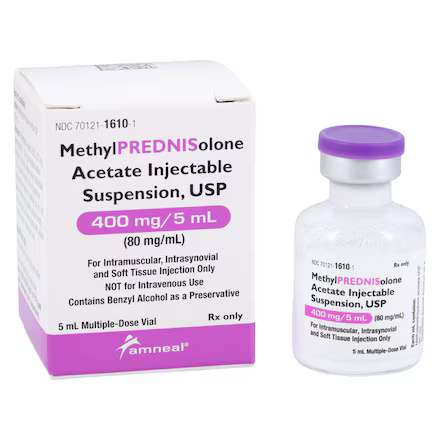
| NDC Number | 70121-1610-01 |
| Active Ingredient | Methylprednisolone Acetate |
| Strength / Concentration | 80 mg / mL (400 mg total per vial) |
| Volume | 5 mL |
| Container Type | Multiple-Dose Vial (MDV) |
| Preservative | Benzyl Alcohol |
| Application | IM / Intra-synovial / Soft Tissue / Intralesional |
| Storage | 20°C to 25°C (68°F to 77°F); Shake well before use |
Key Features
-
Extended Release Mechanism: The acetate ester provides a slow-release profile, offering localized or systemic anti-inflammatory effects that can last for several weeks.
-
Economical Multi-Dose Vial: The 5 mL volume is designed for clinical environments requiring multiple doses or higher volume administrations, featuring a preserved formulation for repeated access.
-
High Concentration (80 mg/mL): Provides a potent dose in a smaller fluid volume, which is particularly advantageous for intra-articular injections into smaller joints.
-
Superior Hemostasis & Stability: Formulated as a homogeneous white suspension that maintains its integrity when stored correctly and shaken prior to administration.
-
Broad Route Versatility: Indicated for deep intramuscular injection, intra-synovial/intra-articular placement, or intralesional therapy.
Clinical Applications
-
Rheumatologic Disorders: Adjunctive therapy for acute gouty arthritis, rheumatoid arthritis, and osteoarthritis.
-
Dermatologic Conditions: Treatment of severe psoriasis, keloids, and localized lichen planus.
-
Allergic States: Management of intractable allergic rhinitis or contact dermatitis.
-
Ophthalmic Inflammation: Used for severe acute and chronic allergic and inflammatory processes involving the eye.
⚠️ Usage Note: SHAKE WELL immediately before use to ensure a uniform suspension. Not for intravenous (IV) use.
⚠️ Safety Alert: Contains benzyl alcohol; do not use in neonates or for epidural/intrathecal administration.
⚠️ Storage: Protect from freezing. Discard the vial 28 days after the initial puncture, or according to your facility’s policy for multi-dose vials.