Product Specifications
| Specification | Details |
| Manufacturer / Brand | Amneal Biosciences LLC |
| Manufacturer # / SKU | 70121116801 |
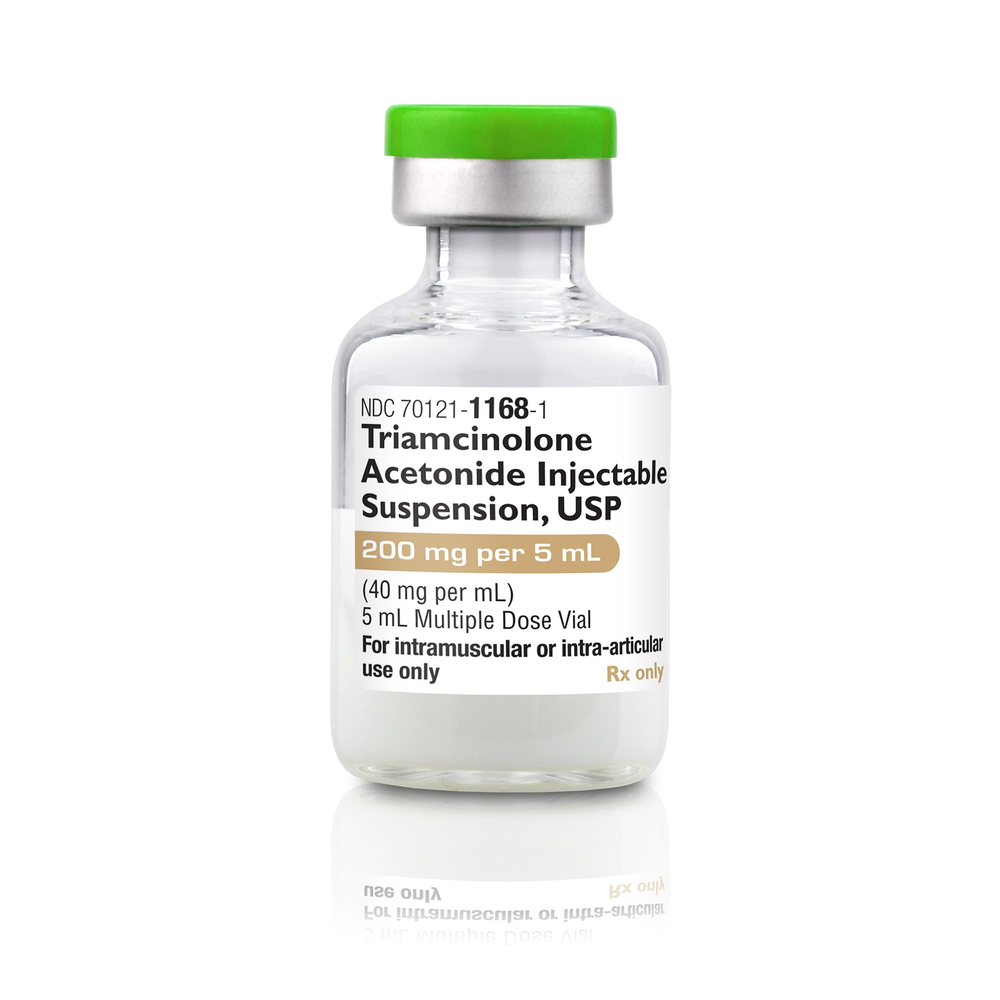
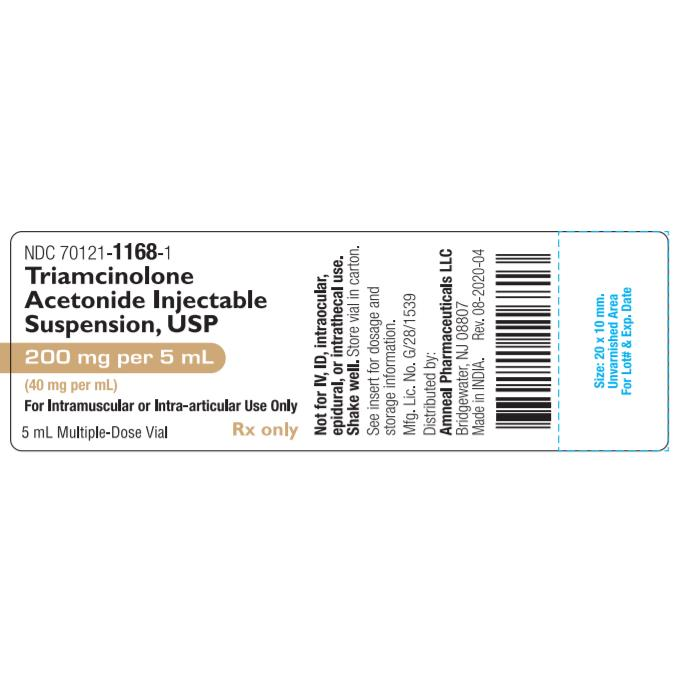
| NDC Number | 70121-1168-01 |
| Active Ingredient | Triamcinolone Acetonide |
| Concentration | 40 mg / mL |
| Total Volume | 5 mL (200 mg total per vial) |
| Container Type | Multi-Dose Vial (MDV) |
| Application | Glucocorticoid / Anti-inflammatory |
| Route of Admin | Intramuscular (IM) / Intra-articular |
| Storage | Store at 20°C to 25°C; Do Not Freeze |
| Packaging / Sold As | Individual 5 mL Vial |
| UNSPSC Code | 51101807 |
Key Features
-
Multi-Dose Efficiency: The 5 mL vial allows for multiple doses to be drawn from a single container, making it a cost-effective choice for busy clinics performing regular joint injections.
-
Extended Duration: Like other triamcinolone suspensions, it provides long-lasting therapeutic effects, often sustained over several weeks from a single injection.
-
Preserved Formula: Contains benzyl alcohol (0.99%) as a preservative, which is standard for multi-dose vials to prevent microbial growth during repeated access.
-
Liquid Suspension: Consists of micronized powder in liquid form; the product appears as an opaque, milky-white liquid.
-
AB-Rated Generic: A therapeutic equivalent to Kenalog®-40, ensuring the same clinical efficacy and safety profile.
Clinical Applications
-
Joint Care: Primarily used for intra-articular injection to treat rheumatoid arthritis, osteoarthritis, and acute bursitis.
-
Systemic Therapy: Administered deep intramuscularly for severe allergic reactions or dermatologic conditions when oral therapy is insufficient.
-
Soft Tissue: Effective for intralesional treatment of keloids and localized inflammatory lesions.
⚠️ Handling Note: As a suspension, the vial must be shaken well before each use to ensure the micronized powder is evenly distributed.
⚠️ Strict Warning: Not for Intravenous (IV) use. The particulate matter in the suspension can cause severe vascular complications if injected into a vein.
⚠️ Safety: Because this is a multi-dose vial, strict aseptic technique must be followed during every withdrawal. Discard the vial according to facility policy (typically 28 days after first puncture) or the manufacturer’s expiration.






Reviews
There are no reviews yet.