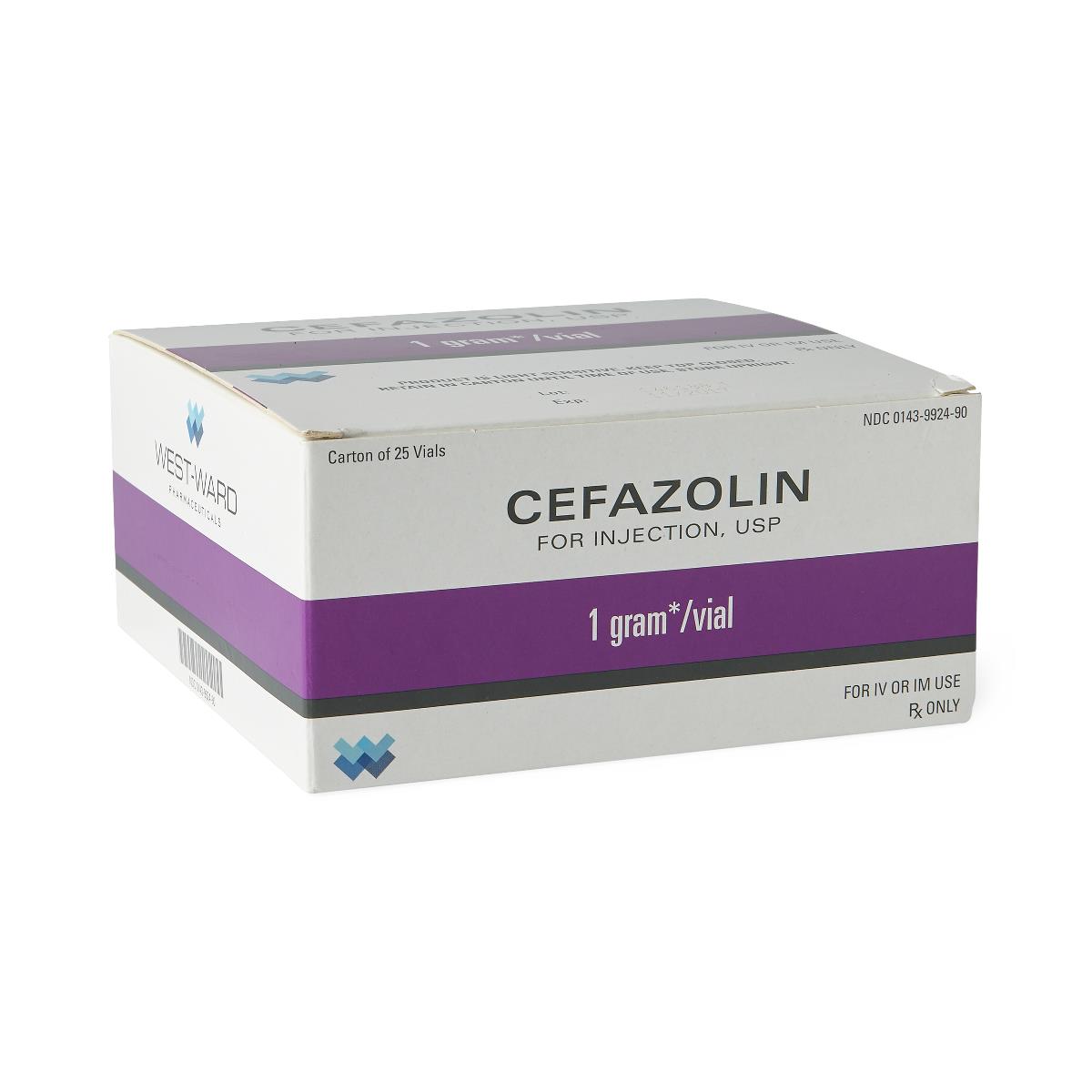
Product Specifications
| Specification | Details |
| Manufacturer / Brand | Hikma Pharmaceuticals USA Inc. |
| Manufacturer # / SKU | 00143-9924-90 |
| NDC Number | 00143-9924-90 |
| Generic Drug Name | Cefazolin Sodium (Preservative-Free) |
| Drug Class | Cephalosporin Antibacterial |
| Strength | 1 Gram |
| Volume / Vial Size | 10 mL Vial (reconstituted to 3 mL) |
| Type | Intramuscular (IM) or Intravenous (IV) |
| Preservative Status | Preservative-Free |
| Container Type | Single-Dose Vial (SDV) |
| Packaging / Sold As | Box of 25 |
| UNSPSC Code | 51282532 |
Key Features
-
Broad-Spectrum Antibiotic: Effectively targets a wide variety of Gram-positive and some Gram-negative bacteria.
-
Versatile Injection Methods: Fully validated for both IV and IM routes, providing flexibility in acute care settings.
-
High-Purity Formulation: A preservative-free sterile powder that ensures patient safety during high-dose or surgical administration.
-
Reconstitution Ready: The 10 mL vial allows for easy aseptic dilution with sterile water or appropriate diluents.
⚠️ Handling: This is a prescription drug (Rx). Contraindicated in patients with a known allergy to cephalosporin antibiotics. Store at controlled room temperature of 20°C to 25°C (68°F to 77°F) and protect from light.





Reviews
There are no reviews yet.