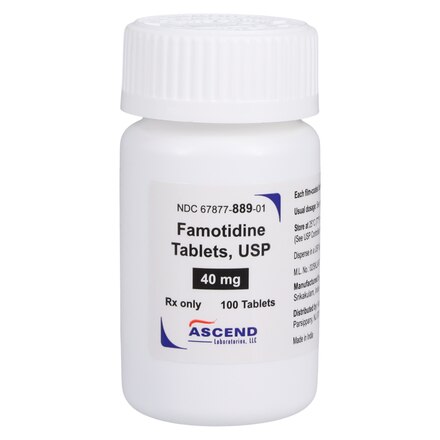
Product Specifications
| Specification | Details |
| Manufacturer / Brand | Ascend Laboratories, LLC |
| Manufacturer # / SKU | 67877088901 |
| NDC Number | 67877-0889-01 |
| Active Ingredient | Famotidine 40 mg |
| Application | Gastric Acid Reducer |
| Dosage Form | Tablet (Film-Coated) |
| Strength | 40 mg |
| Color / Shape | Tan / Round |
| Markings | “A” on one side; “2” on the other |
| Packaging / Sold As | Bottle of 100 Tablets |
| Storage Requirements | USP Controlled Room Temperature 20°C to 25°C |
| UNSPSC Code | 51102710 |
Key Features
-
Maximum Strength Secretion Control: Effectively inhibits both concentration and volume of gastric acid, providing up to 12 hours of relief per dose.
-
Targeted Therapy: The 40 mg strength is the standard dose for the acute treatment of active duodenal and benign gastric ulcers.
-
Stable Bioavailability: Film-coated to ensure consistent dissolution and absorption in the GI tract.
-
Minimal Side Effects: Known for a clean safety profile with significantly fewer drug-to-drug interactions compared to first-generation H2 blockers.
-
Professional Grade: Manufactured under strict USP guidelines for clinical and hospital pharmacy dispensing.
Clinical Applications
-
Active Duodenal/Gastric Ulcers: Short-term treatment (up to 8 weeks) to promote healing of the gastric or duodenal mucosa.
-
Erosive Esophagitis: Diagnosed by endoscopy, the 40 mg dose is used to treat symptoms and help heal the esophagus.
-
Zollinger-Ellison Syndrome: Management of pathological hypersecretory conditions where high-dose acid suppression is vital.
-
Severe GERD: For patients who require more intensive acid management than over-the-counter strengths provide.
⚠️ Usage Note: Dosage should be reduced in patients with severe renal insufficiency (CrCl < 60 mL/min) to avoid central nervous system side effects such as confusion.
⚠️ Safety: Not intended for immediate relief of occasional heartburn; should be taken consistently as prescribed for long-term management.
⚠️ Pregnancy/Lactation: Use only if clearly needed. Famotidine is excreted in human milk; caution should be exercised when administered to a nursing woman.