Product Specifications
| Specification | Details |
| Manufacturer / Brand | Long Grove Pharmaceuticals LLC |
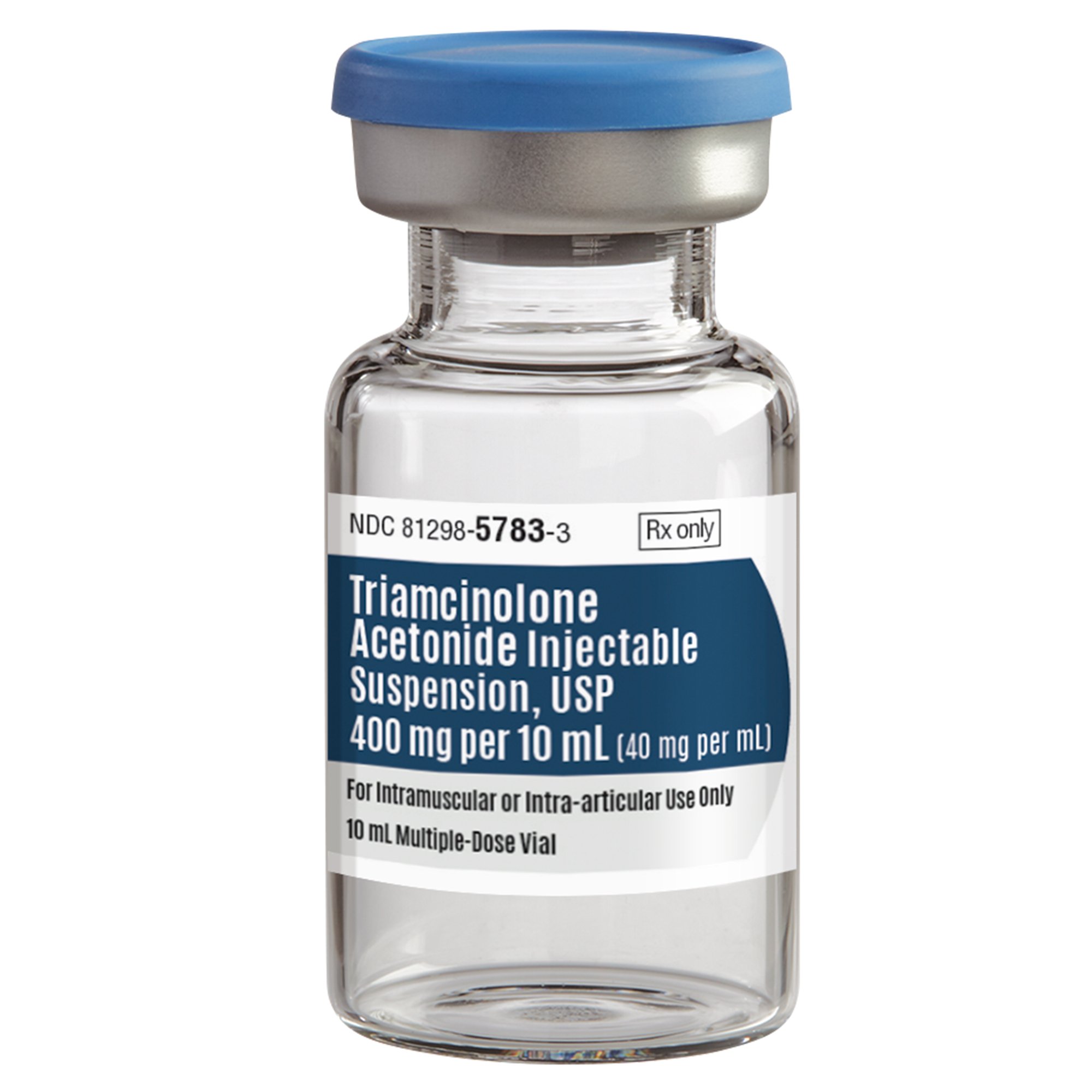
| Manufacturer # / SKU | 81298-5783-3 (81298578303) |
| Product Type | Long-Acting Glucocorticoid / Corticosteroid |
| Active Ingredient | Triamcinolone Acetonide |
| Concentration | 40 mg / mL |
| Total Content | 400 mg per 10 mL vial |
| Dosage Form | Injectable Suspension, USP |
| Container Type | Multiple-Dose Vial (MDV) |
| Administration Route | Intramuscular or Intra-articular Only |
| Equivalent To | Kenalog®-40 |
| Latex Status | Vial stopper not made with natural rubber latex |
| Packaging / Sold As | 1 Vial per Box |
| NDC Number | 81298-5783-03 |
| UNSPSC Code | 51422210 |
Key Features
-
Potent Anti-Inflammatory: Effectively reduces inflammation and pain associated with various chronic and acute conditions.
-
Versatile Application: Approved for both intramuscular (systemic) and intra-articular (localized joint) injections.
-
Generic Value: Provides a high-quality, FDA-approved generic alternative to brand-name Kenalog®-40.
-
Multiple-Dose Efficiency: The 10 mL vial allows for multiple draws, making it a cost-effective choice for clinics managing multiple patients.
-
Safety & Quality: Manufactured with high standards, featuring a latex-free vial stopper to prevent allergic reactions.
⚠️ Contraindications: Not for intravenous, intradermal, or intraocular use. Contains benzyl alcohol; not for use in neonates.