Product Specifications
| Specification | Details |
|---|---|
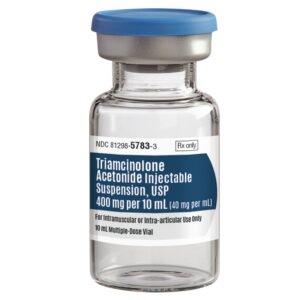
| Manufacturer / Brand | Bristol-Myers Squibb |
| Manufacturer # / SKU | 029328 (NDC 00003-0293-28) |
| Product Type | Triamcinolone Acetonide Injectable Suspension |
| Strength / Concentration | 40 mg/mL |
| Volume | 10 mL Vial (MDV) |
| Dosage Form | Sterile aqueous suspension |
| Route of Administration | Intramuscular or Intra-Articular only |
| Preservative | Contains benzyl alcohol (0.99% w/v) |
| Packaging / Sold As | Each vial (10 mL mDV) |
| Shelf Life | Refer to vial label (typically 36 months) |
| Recommended Storage | Store at 20–25 °C (68–77 °F); protect from light; do not freeze |
| Latex Status | Not made with natural rubber latex |
| UNSPSC Code | 51422210 |
Key Features
-
40 mg/mL dose of triamcinolone acetonide—potent glucocorticoid for IM/IA use
-
Single-vial packaging supports accurate dosing in professional care settings
-
Manufacturer-brand quality from Bristol-Myers Squibb
-
Designed for deep muscle or joint injection (not for IV, epidural, intraocular, or intradermal use)
-
Requires strict aseptic technique and awareness of contraindications (e.g., live vaccines, infections)




Reviews
There are no reviews yet.