Product Specifications
| Specification | Details |
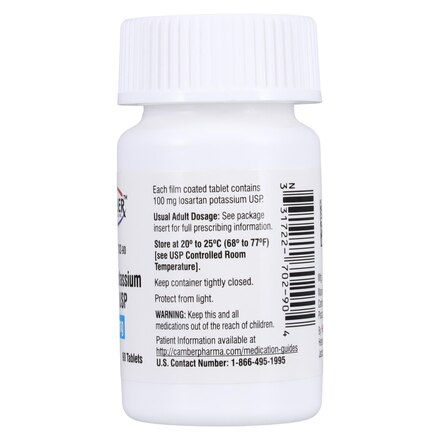
| Manufacturer / Brand | Camber Pharmaceuticals, Inc. |
| Manufacturer # / SKU | 31722070290 |
| NDC Number | 31722-702-90 |
| Active Ingredient | Losartan Potassium 100 mg |
| Application | Antihypertensive / Renal Protection |
| Dosage Form | Tablet (Film-Coated) |
| Strength | 100 mg |
| Color / Shape | White / Oval |
| Markings | “C” on one side and “15” on the other |
| Packaging | Bottle of 90 Tablets |
| Storage Requirements | 20°C to 25°C (68°F to 77°F); keep container tightly closed |
Key Features
-
Maximized Potency: Provides the highest standard therapeutic dose of Losartan for robust 24-hour blood pressure management.
-
Stroke Risk Reduction: Indicated to reduce the risk of stroke in patients with hypertension and left ventricular hypertrophy (LVH).
-
Renal Protective Mechanism: Effectively manages kidney health in patients with Type 2 diabetes by reducing proteinuria and slowing the decline of GFR.
-
Low Incidence of Side Effects: Offers a significantly lower risk of dry cough compared to ACE inhibitors, improving patient compliance.
-
Specific Uricosuric Property: Helps promote the excretion of uric acid, a unique benefit within the ARB class for patients with elevated serum urate.
Clinical Applications
-
Advanced Hypertension: Targeted for patients whose blood pressure is not adequately controlled by 50 mg daily doses.
-
Hypertensive Patients with LVH: Used to mitigate the risk of cardiovascular events and stroke.
-
Diabetic Kidney Disease: Prescribed as a frontline defense against the progression of nephropathy in hypertensive Type 2 diabetics.
⚠️ Usage Note: To maintain stability, ensure the bottle remains tightly sealed. The film coating is designed to protect the hygroscopic core of the tablet.
⚠️ Black Box Warning: FETAL TOXICITY. If pregnancy is detected, discontinue use immediately. Exposure to drugs that act on the renin-angiotensin system during the second and third trimesters can lead to severe fetal injury or death.
⚠️ Safety Alert: Use with extreme caution in patients with hepatic impairment, as plasma concentrations can be significantly elevated. Monitor for signs of hyperkalemia when combined with potassium-sparing diuretics.