Product Specifications
| Specification | Details |
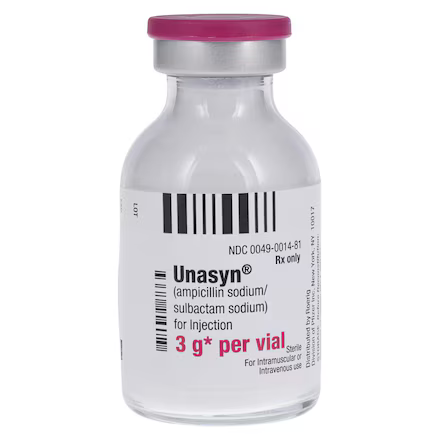
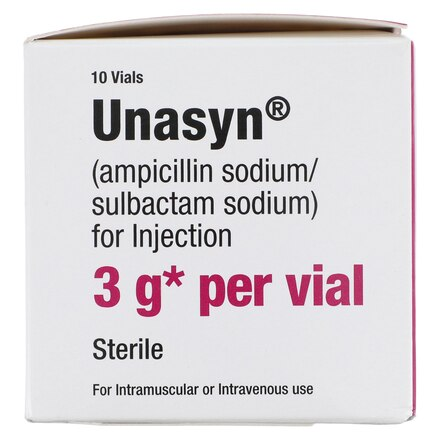
| Manufacturer / Brand | Pfizer Injectables / Unasyn® |
| Manufacturer # / SKU | 00049001483 |
| NDC Number | 00049-0014-83 |
| Active Ingredients | Ampicillin Sodium / Sulbactam Sodium |
| Total Strength | 3 g per Vial (2 g Ampicillin / 1 g Sulbactam) |
| Container Type | Single-Dose Vial (SDV) |
| Formulation | Sterile Powder for Reconstitution |
| Route of Admin | Intravenous (IV) or Intramuscular (IM) |
| Packaging | 10 Vials per Carton |
| Latex Content | Not made with natural rubber latex |
Key Features
-
Beta-Lactamase Protection: Sulbactam effectively “neutralizes” bacterial resistance enzymes, restoring and extending the bactericidal power of ampicillin.
-
Broad Pathogen Coverage: Indicated for infections caused by Staphylococcus aureus, E. coli, Klebsiella species, Proteus mirabilis, Bacteroides fragilis, and Acinetobacter species.
-
Hospital Standard: As a Pfizer brand-name product, it meets the highest standards for purity, stability, and clinical reliability.
-
Dual Administration Paths: Can be given via deep IM injection or as an IV infusion (after further dilution) to suit the patient’s clinical needs.
-
Preservative-Free: Formulated as a sterile dry powder to ensure stability without the need for additional chemical preservatives.
Clinical Applications
-
Skin & Skin Structure Infections: Effective against complex cellulitis and diabetic foot infections.
-
Intra-Abdominal Infections: Used in the management of peritonitis and appendicitis.
-
Gynecological Infections: Treatment for pelvic inflammatory disease (PID) and postpartum infections.
-
Surgical Prophylaxis: Often used to prevent infection during contaminated abdominal or pelvic surgeries.
⚠️ Black Box & Safety Alerts: Severe Anaphylaxis. Serious and occasionally fatal hypersensitivity reactions have been reported. Always verify patient allergy history to penicillins or cephalosporins.
⚠️ Reconstitution Note: Use the reconstituted solution within 1 hour if kept at room temperature, or 4 hours if refrigerated, depending on the diluent used.
⚠️ Safety Alert: Clostridioides difficile-associated diarrhea (CDAD) has been reported with the use of nearly all antibacterial agents, including Unasyn.