Product Specifications
| Specification | Details |
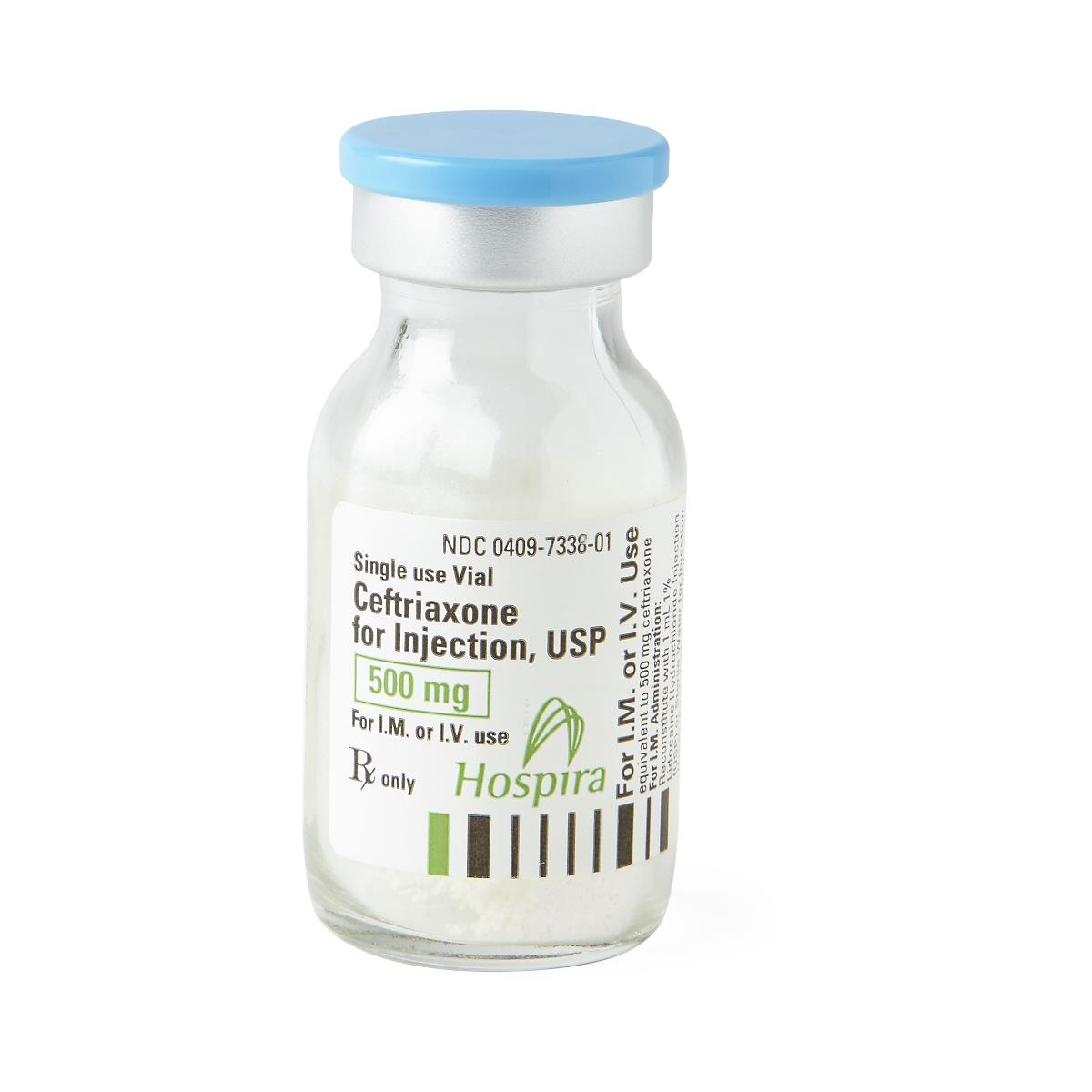
| Manufacturer / Brand | Pfizer / Hospira Worldwide Inc. |
| Manufacturer # / SKU | 00409733801 |
| NDC Number | 00409-7338-01 |
| Generic Drug Name | Ceftriaxone Sodium (Preservative-Free) |
| Drug Class | Cephalosporin Antibacterial |
| Strength | 500 mg |
| Volume / Vial Size | 15 mL Glass Fliptop Vial |
| Type | Intramuscular (IM) or Intravenous (IV) |
| Preservative Status | Preservative-Free |
| Container Type | Single-Dose Vial (SDV) |
| Packaging / Sold As | Box of 10 |
| UNSPSC Code | 51284150 |
Key Features
-
Bactericidal Action: Effectively inhibits bacterial cell wall synthesis, targeting both Gram-positive and Gram-negative pathogens.
-
Versatile Clinical Use: Validated for multiple routes, including slow IV infusion or deep IM injection.
-
Enhanced Safety: Preservative-free and Non-DEHP formulation, not made with natural rubber latex to minimize patient sensitivity.
-
Proven Stability: Supplied as a white to yellowish crystalline powder that maintains high potency until reconstituted.
⚠️ Handling: This is a prescription drug (Rx). Store at controlled room temperature of 20°C to 25°C (68°F to 77°F) and protect from light. Once reconstituted, follow specific labeling instructions for stability and storage of the solution.






Reviews
There are no reviews yet.