Product Specifications
| Specification | Details |
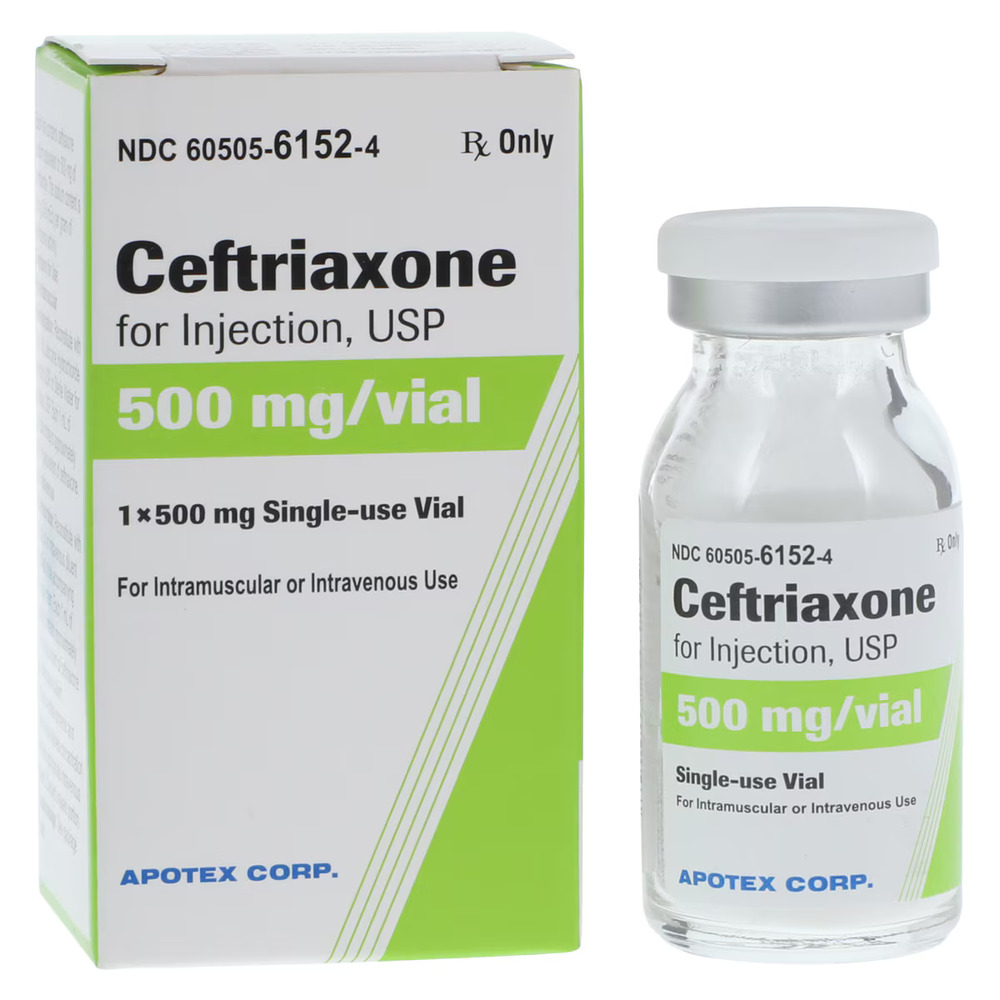
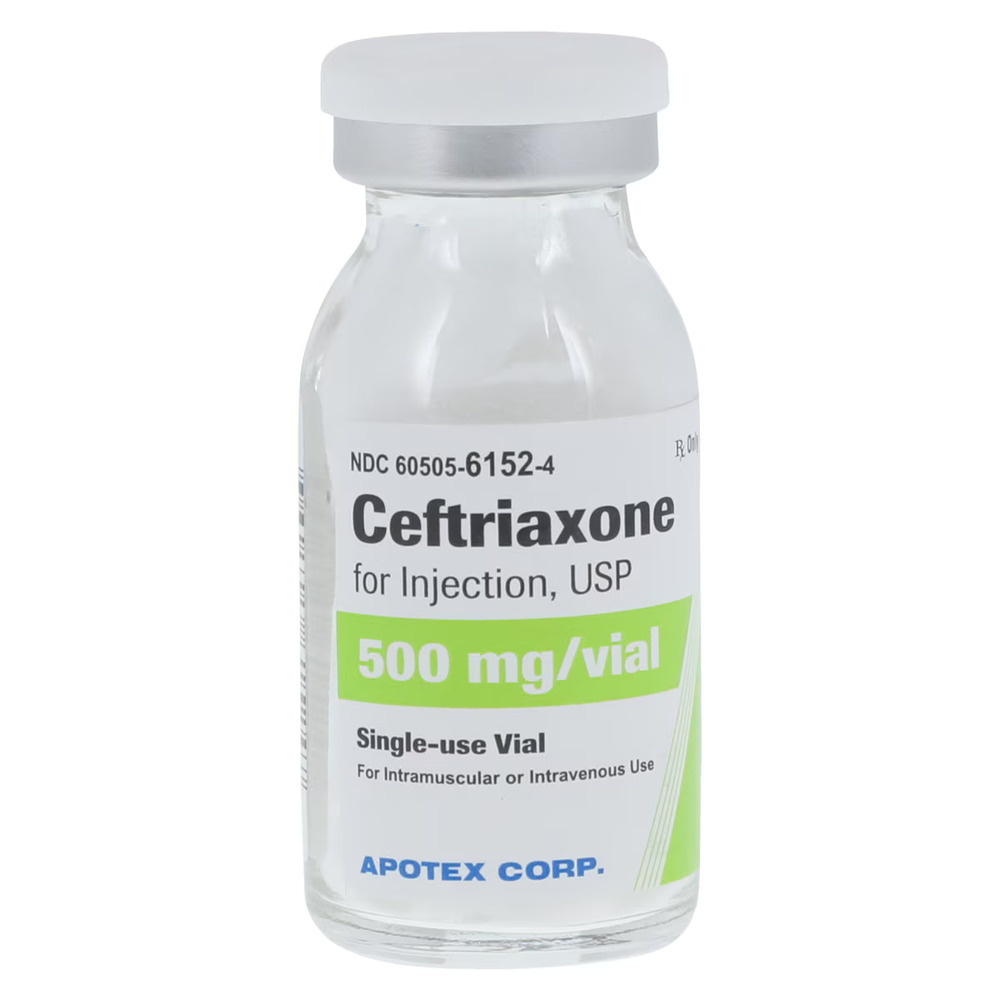
| Manufacturer / Brand | Apotex Corp |
| Manufacturer # / SKU | 60505615(201/204) |
| NDC Number | 60505-6152-01 |
| Generic Drug Name | Ceftriaxone Sodium (Preservative-Free) |
| Drug Class | Cephalosporin Antibacterial |
| Strength | 500 mg |
| Volume / Vial Size | 10 mL Vial |
| Type | Intramuscular (IM) or Intravenous (IV) |
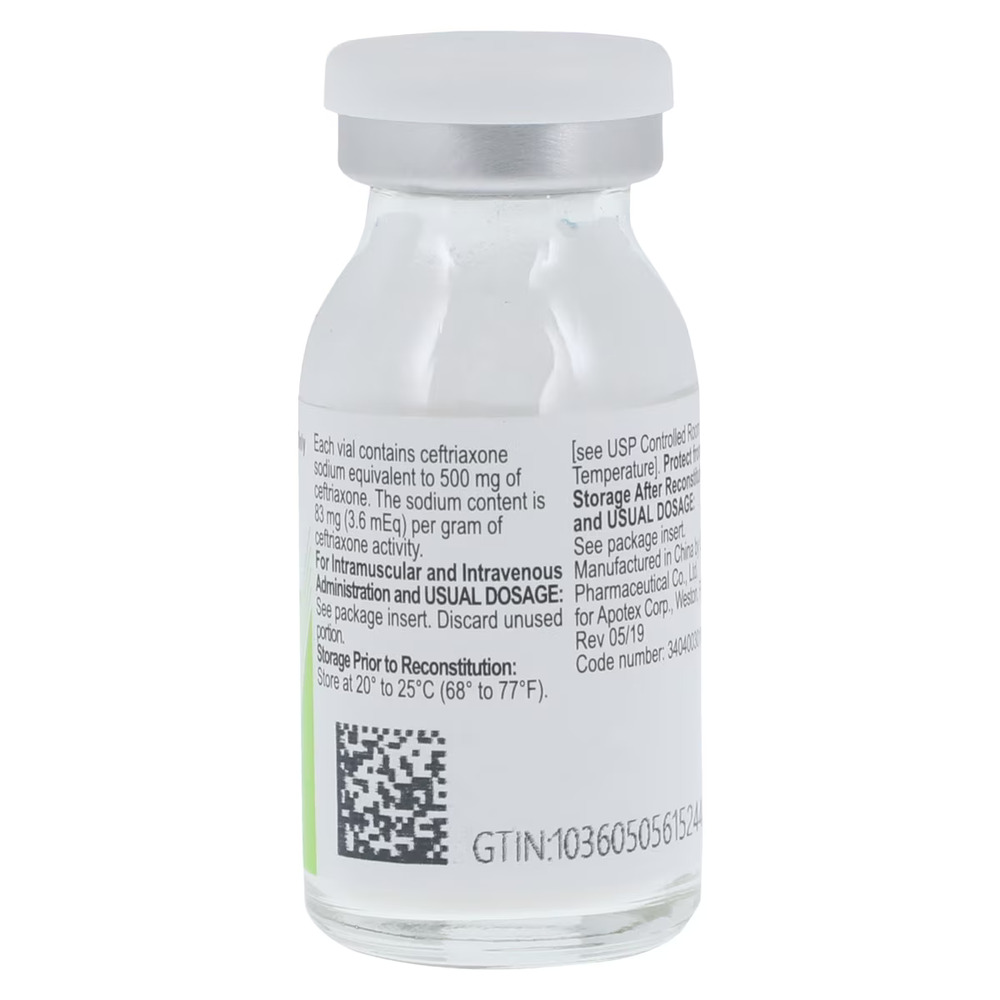
| Preservative Status | Preservative-Free |
| Container Type | Single-Dose Vial (SDV) |
| Packaging / Sold As | Single Vial / Box of 10 |
| UNSPSC Code | 51284150 |
Key Features
-
Bactericidal Action: Effectively targets Gram-positive and Gram-negative bacteria by inhibiting bacterial cell wall synthesis.
-
Versatile Delivery: Validated for both deep IM injection and slow IV infusion, providing options for varied clinical environments.
-
High Purity Formulation: A preservative-free, dye-free, and latex-free sterile powder designed to meet strict USP standards for patient safety.
-
Reconstitution Ready: Supplied as a crystalline powder in glass vials to maintain stability and potency until the point of care.
⚠️ Handling: This is a prescription drug (Rx). Use should be guided by culture and susceptibility testing to reduce the development of drug-resistant bacteria. Store at controlled room temperature of 20°C to 25°C (68°F to 77°F) and protect from light.