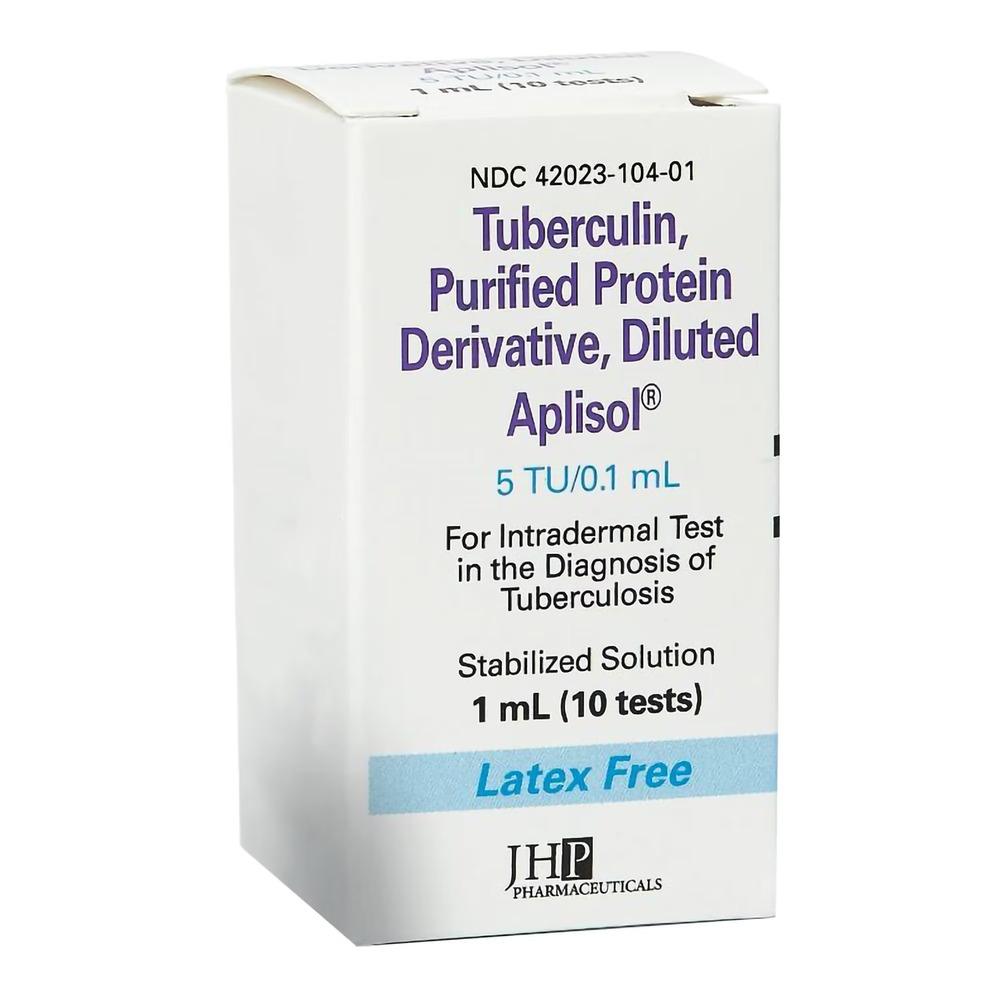
Product Specifications
| Specification | Details |
| Manufacturer / Brand | Endo USA, Inc. / Aplisol® |
| Manufacturer # / SKU | 42023010401 |
| NDC # | 42023-0104-01 |
| Product Type | Tuberculosis Skin Test Antigen |
| Strength | 5 TU (Tuberculin Units) per 0.1 mL |
| Volume | 1 mL (10 Tests per Vial) |
| Container Type | Multi-Dose Vial (MDV) |
| Administration Route | Intradermal Only (Mantoux Method) |
| Sterility | Sterile, Stabilized Solution |
| Latex Content | Not Made with Natural Rubber Latex |
| Packaging / Sold As | Individual 1 mL Vial |
| UNSPSC Code | 51201651 |
Key Features
-
Standardized Diagnostic Potency: Bioequivalent to the standard PPD-S of the U.S. Public Health Service, ensuring consistent and reliable skin test results across different clinical settings.
-
Ready-to-Use Formulation: Provided as a diluted, stabilized solution that requires no further processing, reducing the risk of contamination and dosage errors during preparation.
-
Multi-Dose Efficiency: The 1 mL vial configuration provides 10 tests, making it a cost-effective solution for large-scale employee health screenings and clinical tuberculosis surveillance.
-
Clear Reaction Interpretation: Specifically designed for the Mantoux test, where the area of induration (not erythema) is measured 48 to 72 hours post-injection to determine infection status.
-
Preservative Stabilized: Contains 0.25% phenol as a preservative to maintain solution integrity over the life of the multi-dose vial.






Reviews
There are no reviews yet.