Product Specifications
| Specification | Details |
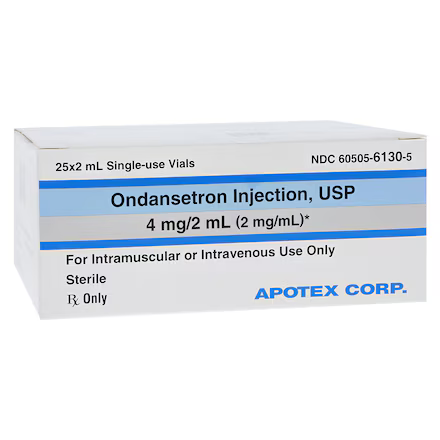
| Manufacturer / Brand | Apotex Corp |
| Manufacturer # / SKU | 60505613005 |
| NDC Number | 60505-6130-05 |
| Active Ingredient | Ondansetron Hydrochloride |
| Strength | 4 mg / 2 mL (2 mg / mL) |
| Volume per Unit | 2 mL |
| Container Type | Multiple-Dose Vial (MDV) |
| Formulation | Sterile solution with Methylparaben/Propylparaben |
| Route of Admin | Intravenous (IV) or Intramuscular (IM) |
| Packaging | 25 Vials per Carton |
| Latex Content | Not made with natural rubber latex |
Key Features
-
Proven Efficacy: Gold standard therapy for managing highly emetogenic chemotherapy-induced nausea and vomiting (CINV).
-
Multiple-Dose Convenience: The MDV format is ideal for clinical settings where weight-based pediatric dosing or lower adult doses are required across multiple patients.
-
Rapid Action: Provides fast onset of anti-emetic effects when administered via IV bolus or IM injection.
-
Preserved Formula: Contains antimicrobial preservatives to maintain sterility during multiple entries into the vial.
-
Cost-Effective Generic: Offers the same therapeutic efficacy as brand-name Zofran® at a more accessible price point for healthcare facilities.
Clinical Applications
-
Chemotherapy Support: Prevention of nausea and vomiting associated with initial and repeat courses of emetogenic cancer therapy.
-
Surgical Recovery: Prevention and treatment of postoperative nausea and vomiting (PONV) in adult and pediatric patients.
-
Radiation Therapy: Management of nausea in patients receiving total body irradiation or localized abdominal radiation.
-
Off-Label Use: Often used in emergency departments for intractable vomiting associated with gastroenteritis.
⚠️ Safety Alert: QT Prolongation. Ondansetron can cause dose-dependent prolongation of the QT interval. Avoid in patients with congenital long QT syndrome.
⚠️ Usage Note: For IV administration, it may be given undiluted over 2 to 5 minutes or diluted in 50 mL of compatible fluid and infused over 15 minutes.
⚠️ Contraindications: Contraindicated in patients with known hypersensitivity to ondansetron or any component of the formulation. Concomitant use with apomorphine is contraindicated.