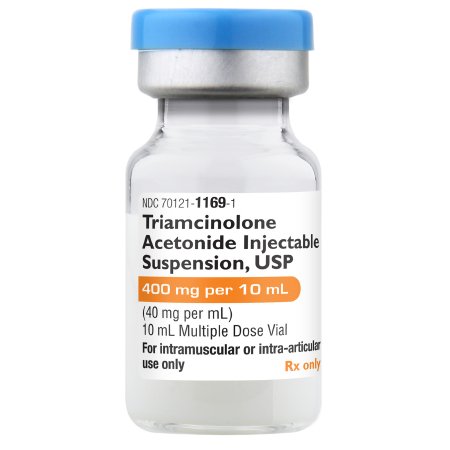
Product Specifications
| Specification | Details |
| Manufacturer / Brand | Amneal Pharmaceuticals, LLC |
| Manufacturer # / SKU | 70121116901 |
| NDC Number | 70121-1169-01 |
| Active Ingredient | Triamcinolone Acetonide |
| Concentration | 40 mg / mL (400 mg per vial) |
| Volume | 10 mL |
| Container Type | Multiple-Dose Vial (MDV) |
| Preservative | Benzyl Alcohol |
| Application | IM / Intra-articular / Intralesional |
| Storage | 20°C to 25°C (68°F to 77°F); Do not freeze |
Key Features
-
Extended Therapeutic Window: Formulated as a microcrystalline suspension to provide a slow-release effect, often delivering anti-inflammatory benefits for 3 to 4 weeks.
-
Versatile Multi-Dose Format: The 10 mL vial is ideal for high-volume clinics, containing a preservative that allows for repeated aseptic withdrawals.
-
Potent Corticosteroid Activity: Provides approximately 5 times the anti-inflammatory potency of hydrocortisone with minimal mineralocorticoid (salt-retaining) activity.
-
Surgical Precision: The suspension is engineered for smooth passage through fine-gauge needles, facilitating accurate placement in joint spaces or soft tissue.
-
Cost-Effective Clinical Supply: Offers a high volume of medication in a single unit, reducing the need for multiple single-dose vials during a busy clinic day.
Clinical Applications
-
Rheumatologic Management: Used for intra-articular injection to treat osteoarthritis, rheumatoid arthritis, and acute bursitis flare-ups.
-
Dermatological Therapy: Effective for intralesional treatment of keloids, discoid lupus erythematosus, and localized hypertrophic lesions.
-
Systemic Allergic Control: Deep intramuscular injection for severe hay fever, asthma, or drug hypersensitivity reactions.
-
Endocrine Disorders: Used in the management of primary or secondary adrenocortical insufficiency.
⚠️ Usage Note: SHAKE WELL BEFORE USE to ensure the suspension is uniform. Not for intravenous (IV), intrathecal, or epidural use.
⚠️ Safety Alert: Use strict aseptic technique when entering the multi-dose vial. Contains benzyl alcohol; do not use in neonates or infants.
⚠️ Storage: Protect from light and store upright. Discard the vial 28 days after initial puncture according to CDC/USP <797> guidelines.


Reviews
There are no reviews yet.