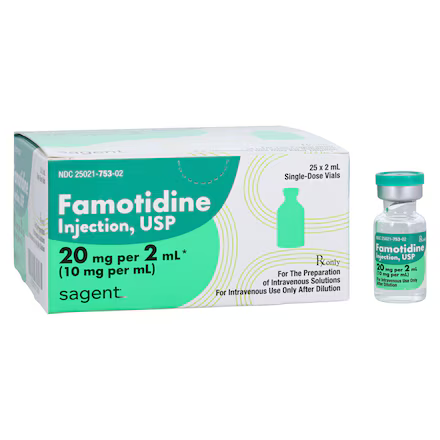
Product Specifications
| Specification | Details |
| Manufacturer / Brand | Sagent Pharmaceuticals |
| Manufacturer # / SKU | 25021-0753-02 |
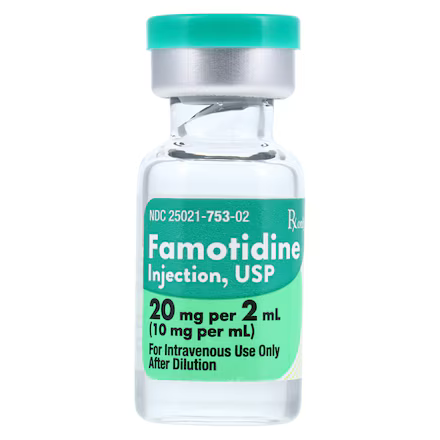
| NDC Number | 25021-0753-02 |
| Active Ingredient | Famotidine 10 mg / mL |
| Application | Histamine H2-Antagonist |
| Dosage Form | Injection, Solution |
| Strength | 20 mg per 2 mL (10 mg/mL) |
| Container Type | Single-Dose Vial |
| Preservative Status | Preservative-Free |
| Storage Requirements | Store at 20 C to 25 C (68 F to 77 F) |
| Packaging / Sold As | Case of 25 Vials |
| UNSPSC Code | 51171902 |
Key Features
-
Preservative-Free Formula: Formulated without preservatives, making it suitable for patients with sensitivities and ideal for single-use clinical applications.
-
Gastric Acid Suppression: Highly effective at reducing the production of stomach acid, aiding in the healing of ulcers and the management of acid reflux.
-
Clinical Versatility: Used for the treatment of active duodenal ulcers and benign gastric ulcers, as well as the maintenance of healed ulcers in adult patients.
-
Standard Potency: Provides 10 mg per mL, allowing for straightforward dosing and administration of the common 20 mg clinical dose.
-
Sagent Safety Labeling: Features the Sagent “PreventIV” labeling and packaging design to help reduce medication errors through clear identification of drug name and strength.
Usage & Administration
-
Administration: For intravenous use only. It may be administered as a slow bolus injection over at least 2 minutes or as a short-term infusion over 15 to 30 minutes.
-
Dilution: For infusion, the 20 mg dose (2 mL) should be diluted with 0.9% Sodium Chloride Injection or 5% Dextrose Injection to a volume of 50 mL or 100 mL.
-
Transition to Oral: Injectable therapy should be discontinued as soon as the patient is able to transition to oral famotidine tablets or suspension.
-
Visual Inspection: Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration.
⚠️Caution: Do not use if the solution is discolored or contains a precipitate.
⚠️Renal Impairment: Because famotidine is primarily excreted by the kidneys, dosage intervals may need to be adjusted (e.g., 20 mg every 36 to 48 hours) in patients with moderate to severe renal insufficiency.
⚠️Storage: Unlike some other brands, the Sagent formulation is generally stored at controlled room temperature. Protect from light by keeping the vials in the original carton until use.




Reviews
There are no reviews yet.