Product Specifications
| Specification | Details |
| Manufacturer / Brand | Rising Pharmaceuticals, Inc. |
| Manufacturer # / SKU | 57237007530 |
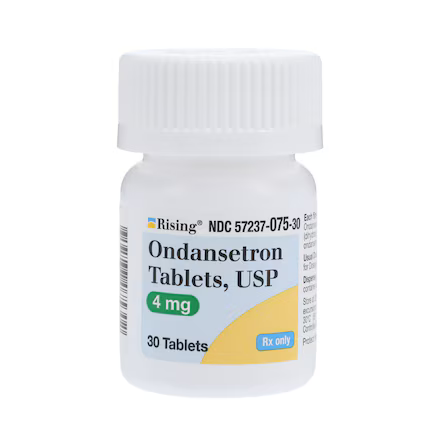
| NDC Number | 57237-0075-30 |
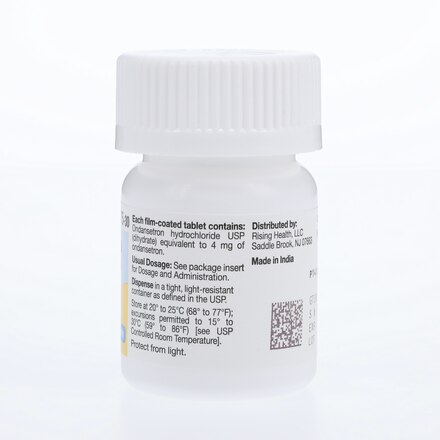
| Active Ingredient | Ondansetron Hydrochloride |
| Strength | 4 mg |
| Dosage Form | Film-Coated Tablet (Swallowable) |
| Color / Shape | White / Oval |
| Packaging | 30 Tablets per Bottle |
| Storage | 20°C to 25°C (68°F to 77°F) |
Key Features
-
Standard Oral Delivery: Ideal for patients who prefer a traditional swallowable tablet over a dissolving ODT or for those who do not like the sweeteners/flavors found in ODT formulations.
-
Stable Bioavailability: Provides consistent systemic absorption for the prevention of CINV (chemotherapy-induced nausea and vomiting).
-
Film-Coated: The smooth coating makes the tablet easier to swallow and masks the bitter taste of the active ingredient.
-
Precise Dosing: The 30-count bottle is a convenient unit-of-use size for a typical post-operative recovery or a single chemotherapy cycle.
-
Cost-Efficient: A reliable generic equivalent to brand-name Zofran® tablets.
Clinical Applications
-
Moderately Emetogenic Chemotherapy: Prevention of nausea and vomiting associated with initial and repeat courses.
-
Radiotherapy: Prevention of nausea in patients receiving total body irradiation or daily fractions to the abdomen.
-
Postoperative Nausea (PONV): Prevention of nausea following surgical procedures where oral intake is tolerated.
-
Off-Label Gastroenteritis: Used in clinical settings to manage vomiting and support oral rehydration therapy.
⚠️ Usage Note: These tablets must be swallowed whole with water. Do not crush or chew unless specifically directed by a healthcare provider.
⚠️ Safety Alert: QT Prolongation. Use with caution in patients with electrolyte abnormalities or those taking other medications that can affect heart rhythm.
⚠️ Side Effects: Common reactions include headache, constipation, and a sensation of warmth or flushing. Monitor for signs of a severe allergic reaction or Serotonin Syndrome.