Product Specifications
| Specification | Details |
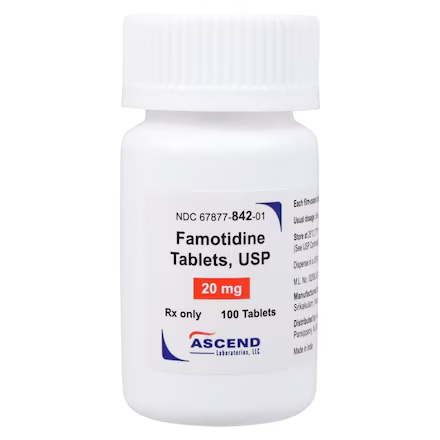
| Manufacturer / Brand | Ascend Laboratories, LLC |
| Manufacturer # / SKU | 67877084201 |
| NDC Number | 67877-0842-01 |
| Active Ingredient | Famotidine 20 mg |
| Application | Gastric Acid Reducer |
| Dosage Form | Tablet (Film-Coated) |
| Strength | 20 mg |
| Color / Shape | Beige / Round |
| Markings | “A” on one side; “1” on the other |
| Packaging / Sold As | Bottle of 100 Tablets |
| Storage Requirements | USP Controlled Room Temperature 20°C to 25°C |
| UNSPSC Code | 51102710 |
Key Features
-
Potent Acid Suppression: Significantly reduces basal and nocturnal gastric acid secretion, as well as secretion stimulated by food and pentagastrin.
-
Rapid Symptom Relief: Onset of action typically occurs within one hour of oral administration, with effects lasting up to 10 to 12 hours.
-
Small, Easy-to-Swallow Tablets: The film-coated round tablets are designed for patient comfort and compliance.
-
Low Drug Interaction Profile: Unlike some other H2 blockers (like cimetidine), famotidine does not interfere with the cytochrome P450 enzyme system, making it safer for patients on multiple medications.
-
Clinical Standard: A primary choice for treating active ulcers and preventing their recurrence.
Clinical Applications
-
Duodenal Ulcers: Short-term treatment of active duodenal ulcers and maintenance therapy at reduced dosage after healing.
-
Gastric Ulcers: Short-term treatment (up to 8 weeks) of benign active gastric ulcers.
-
Gastroesophageal Reflux Disease (GERD): Short-term treatment of patients with symptoms of GERD and for the treatment of erosive esophagitis.
-
Pathological Hypersecretory Conditions: Management of conditions such as Zollinger-Ellison Syndrome.
⚠️ Usage Note: For patients with moderate or severe renal insufficiency, the dose may need to be adjusted by a physician to prevent accumulation of the drug.
⚠️ Safety: While generally well-tolerated, rare side effects may include headache, dizziness, constipation, or diarrhea.
⚠️ Interaction Warning: Use of H2 blockers may reduce the absorption of certain drugs that require an acidic stomach environment, such as ketoconazole or iron salts.