Product Specifications
| Specification | Details |
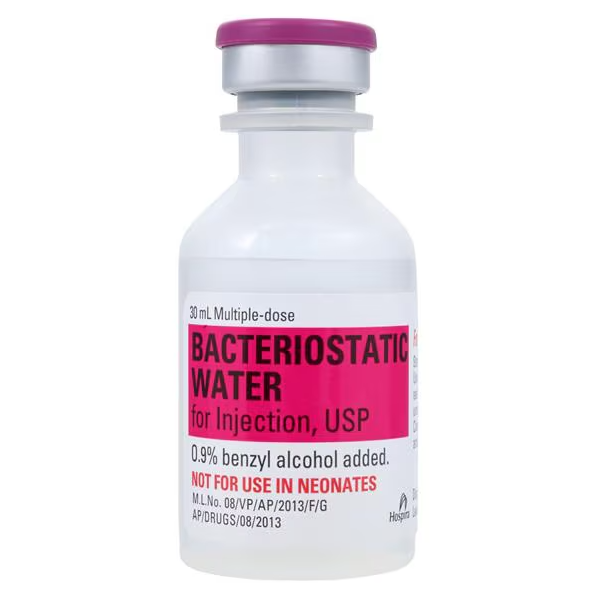
| Manufacturer / Brand | Pfizer Injectables (Hospira) |
| Manufacturer # / SKU | 00409397703 |
| NDC Number | 00409-3977-03 |
| Active Ingredient | Sterile Water for Injection, USP (H2O) |
| Preservative | 0.9% Benzyl Alcohol |
| Volume per Unit | 30 mL |
| Container Type | Multiple-Dose Vial (MDV) |
| Formulation | Preserved / Nonpyrogenic |
| Packaging | 25 Vials per Tray/Box |
| Latex Content | Not made with natural rubber latex |
Key Features
-
Multiple-Dose Access: The bacteriostatic preservative inhibits the growth of most potentially contaminating bacteria, allowing the vial to be used for multiple draws over a 28-day period (once opened).
-
Flip-Top Convenience: Equipped with a protective plastic flip-top cap that ensures the sterile rubber stopper remains uncontaminated until the first use.
-
Large Volume: The 30 mL vial size is ideal for high-volume clinics or pharmacies that perform frequent medication reconstitutions.
-
Standard Compatibility: Meets all USP requirements for pH, particulate matter, and sterility for parenteral diluents.
Clinical Applications
-
Medication Reconstitution: Used to dissolve lyophilized powders into liquid form for injection.
-
Drug Dilution: Used to adjust the concentration of liquid medications to meet specific patient dosage requirements.
-
Pharmacy Compounding: Often utilized in cleanroom environments for the preparation of sterile injectable batches.
⚠️ Black Box & Safety Alert: NOT FOR USE IN NEONATES. Benzyl alcohol has been associated with “Gasping Syndrome” (a fatal toxicity) in newborns and premature infants. Use only preservative-free diluents for this population.
⚠️ Usage Note: When using to dilute drugs, ensure the drug is compatible with the preservative (Benzyl Alcohol). Check the medication’s package insert for specific diluent requirements.
⚠️ Discard Policy: Once the vial has been entered, it must be labeled with the date and discarded within 28 days or as per facility protocol, whichever is sooner.





Reviews
There are no reviews yet.