Product Specifications
| Specification | Details |
| Manufacturer / Brand | Wondfo USA / SAFElife™ T-Cup® |
| Manufacturer # / SKU | TDOA-8104 |
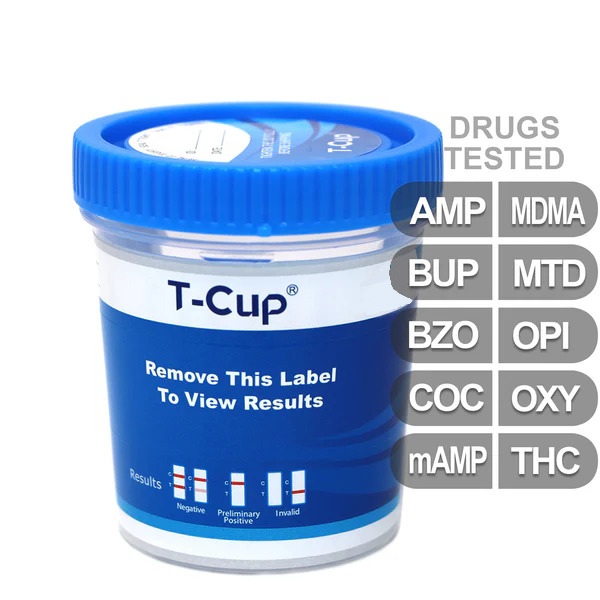
| Test Configuration | 10-Panel All-In-One Cup |
| Panels & Cutoffs | AMP1000, BUP10, BZO300, COC300, mAMP1000, MDMA500, MTD300, OPI2000, OXY100, THC50 |
| Regulatory Status | FDA 510(k) Cleared / CLIA Waived |
| Specimen Type | Human Urine |
| Results Time | 5 Minutes (Stable up to 60 min) |
| Accuracy | >99% (Correlated with GC/MS) |
| Temperature Strip | Integrated (Range: 90°F – 100°F) |
| Sterility | Non-Sterile |
| Shelf Life | 24 Months from Manufacture |
| Storage Requirements | 39°F – 86°F (4°C – 30°C) |
| Packaging / Sold As | Box of 25 Cups |
| UNSPSC Code | 41116142 |
Key Features
-
10-Panel Comprehensive Screen: Detects a broad range of substances including Amphetamines, Buprenorphine, Benzodiazepines, Cocaine, Methamphetamines, MDMA (Ecstasy), Methadone, Opiates, Oxycodone, and Marijuana (THC).
-
Integrated Specimen Validation: Features a built-in temperature strip to immediately confirm the specimen was freshly voided, helping to prevent sample substitution or tampering.
-
Streamlined “One-Step” Design: The collection and testing occur within the same device, minimizing specimen handling and reducing the risk of exposure to biohazards.
-
Easy-to-Read Interpretation: Utilizes high-contrast rose-pink bands to indicate results; the presence of two lines (Control and Test) indicates a negative result, while a single Control line indicates a preliminary positive.