Product Specifications
| Specification | Details |
|---|---|
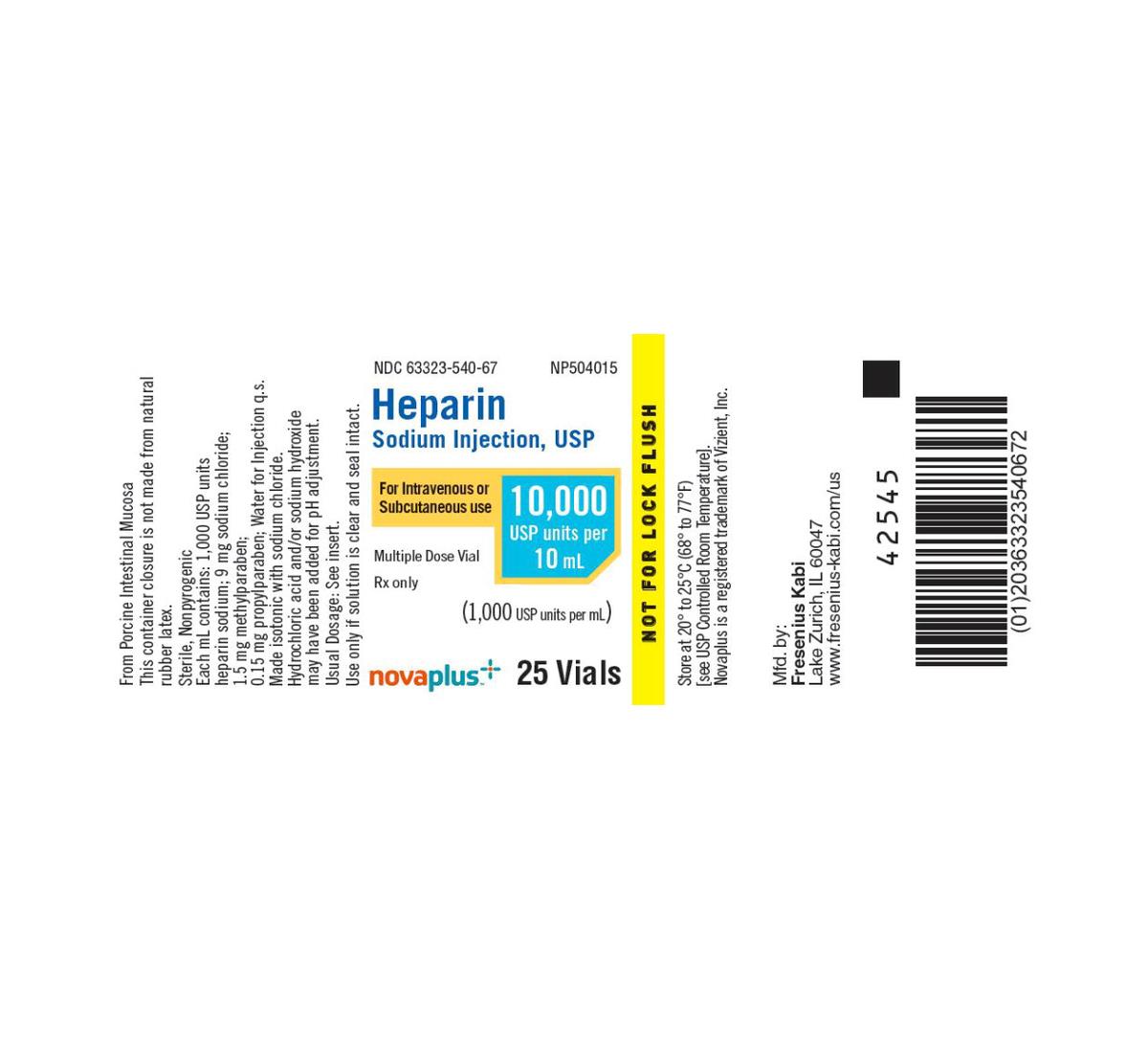
| Manufacturer | Fresenius Kabi USA, LLC |
| Manufacturer # / NDC | 63323‑540‑67 |
| Product Name | Heparin Sodium Injection, USP |
| Strength | 1,000 U/mL |
| Volume per Vial | 10 mL (Multi-Dose) |
| Total Units per Vial | 10,000 Units |
| Container Type | Clear glass multi-dose vial with flip-top closure |
| Tray Quantity | 25 vials per tray |
| Preservative | Contains parabens (methylparaben & propylparaben) |
| Sterility & Pyrogenicity | Sterile, nonpyrogenic; latex-free closure |
| Route of Administration | Intravenous or Subcutaneous |
| Inactive Ingredients | 9 mg sodium chloride, parabens; pH adjusted with HCl/NaOH |
| Storage Conditions | 20–25 °C (USP controlled room temperature); protect from excessive heat |
| Taxable | Yes |
| UNSPSC Code | 51313000 (Anticoagulants) |
Key Features
-
Standard Strength for Anticoagulation: 1,000 U/mL concentration ideal for common IV and SC therapeutic protocols.
-
Convenient Multi‑Dose Format: 10 mL vials with tray-pack of 25 for bulk usage in hospitals and clinics.
-
Paraben Preservation: Contains methyl- and propylparaben for multi-dose stability and microbial protection.
-
Sterile, Latex-Free Closure: Safe for patients with latex sensitivities and meets USP sterility standards.
-
Clear Usage Guidelines: Suitable for IV and SC administration with accurate dosage delivery.
-
Reliable Brand & Traceability: Manufactured by Fresenius Kabi with clear NDC coding and US-based compliance.