Product Specifications
| Specification | Details |
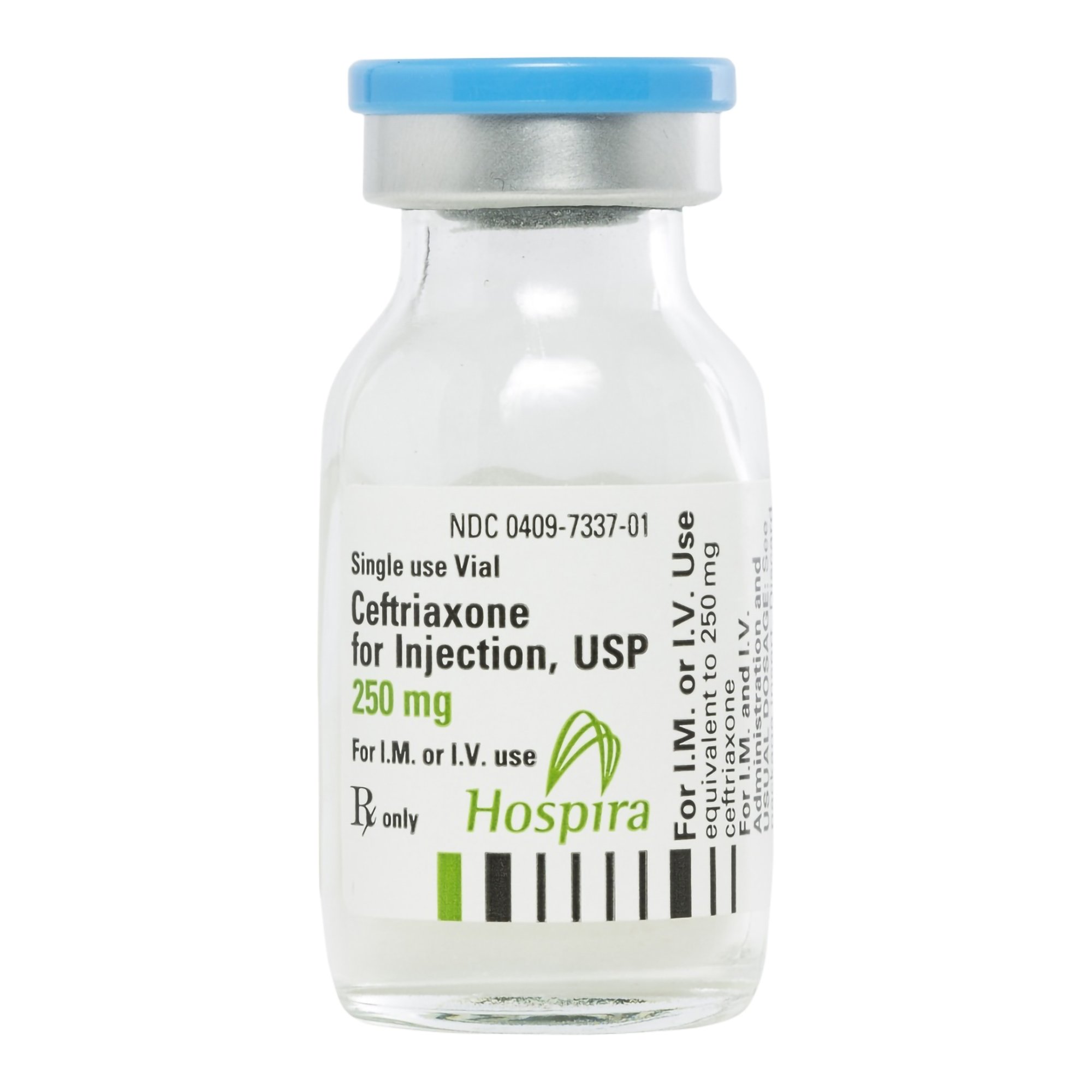
| Manufacturer / Brand | Pfizer / Hospira Worldwide Inc. |
| Manufacturer # / SKU | 00409733701 |
| NDC Number | 00409-7337-01 |
| Generic Drug Name | Ceftriaxone Sodium (Preservative-Free) |
| Drug Class | Cephalosporin Antibacterial |
| Strength | 250 mg |
| Volume / Vial Size | 15 mL Vial |
| Type | Intramuscular (IM) or Intravenous (IV) |
| Preservative Status | Preservative-Free |
| Container Type | Single-Dose Vial (SDV) |
| Packaging / Sold As | Box of 10 |
| UNSPSC Code | 51284150 |
Key Features
-
Targeted Potency: Effective against a broad spectrum of Gram-positive and Gram-negative organisms by inhibiting cell wall synthesis.
-
Flexible Administration: Validated for both deep IM injection and slow IV infusion, allowing for versatile clinical use.
-
High Purity: A preservative-free sterile powder that meets strict USP standards for patient safety and compatibility.
-
Reliable Packaging: Single-dose glass vials ensure dose accuracy and maintain sterility for each administration.
⚠️ Handling: This is a prescription drug (Rx). Use only for infections proven or strongly suspected to be caused by susceptible bacteria. Store at controlled room temperature of 20°C to 25°C (68°F to 77°F) and protect from light.




Reviews
There are no reviews yet.