Product Specifications
| Specification | Details |
| Manufacturer / Brand | Hikma Pharmaceuticals USA |
| Manufacturer # / SKU | 00143957710 |
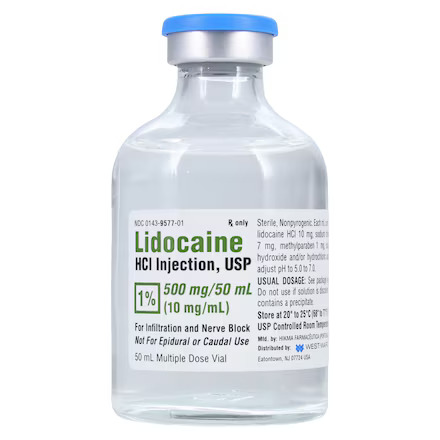
| NDC Number | 00143-9577-10 |
| Active Ingredient | Lidocaine Hydrochloride |
| Strength | 1% (10 mg / mL) |
| Volume per Unit | 50 mL |
| Container Type | Multiple-Dose Vial (MDV) |
| Formulation | Contains Methylparaben (Preserved) |
| Storage Temperature | 20°C to 25°C (68°F to 77°F) |
| Packaging | 10 Vials per Carton |
| Latex Content | Not made with natural rubber latex |
Key Features & Clinical Applications
-
Versatile Anesthesia: Indicated for percutaneous infiltration, peripheral nerve blocks (e.g., brachial plexus, intercostal), and central neural techniques (epidural and caudal blocks).
-
Multiple-Dose Format: The inclusion of methylparaben provides antimicrobial protection, making it ideal for procedures requiring varying volumes across multiple administrations.
-
Rapid Onset: Provides fast-acting local anesthesia with a medium duration of action (typically 1 to 2 hours).
-
Standard Concentration: The 1% (10 mg/mL) concentration is the clinical standard for most infiltration and nerve block procedures.
-
Reliable Generic: Recognized as a high-quality, bioequivalent generic to Xylocaine®.
⚠️ Usage Note: Not for Epidural or Caudal Use. Formulations containing preservatives (like methylparaben) must not be used for spinal or epidural anesthesia, or for any route of administration that would introduce the solution into the cerebrospinal fluid.
⚠️ Safety Alert: Systemic Toxicity. Injections should be made slowly with frequent aspiration to avoid accidental intravascular injection, which can lead to central nervous system toxicity or cardiovascular collapse.
⚠️ Emergency Readiness: Local anesthetics should only be administered by clinicians well-versed in the management of dose-related toxicity. Ensure immediate availability of oxygen and resuscitative drugs.