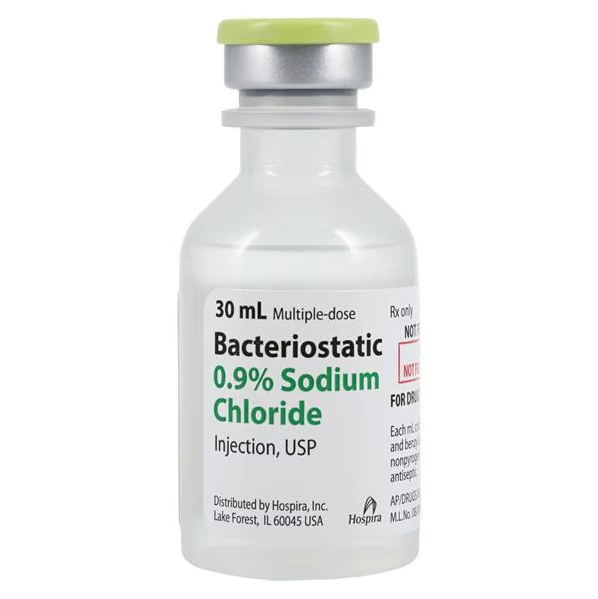
Pfizer Bacteriostatic 0.9% Sodium Chloride Injection, USP, is a sterile, nonpyrogenic, and isotonic solution of sodium chloride in water for injection, intended for use as a diluent or solvent for the parenteral administration (IV, IM, or SC) of drugs. The solution contains 0.9% (9 mg/mL) Benzyl Alcohol added as a bacteriostatic preservative, which allows for multiple withdrawals from the vial.
⚠️ WARNING: Solutions containing benzyl alcohol are contraindicated for use in neonates due to the potential for toxicity. Preservative-free Sodium Chloride Injection should be used for this patient population.
Product Specifications
| Specification | Details |
| Manufacturer / Brand | Pfizer |
| Manufacturer # / SKU | 00409-1966-07 (00409196607) |
| Product Type | Solution / Diluent (Rx) |
| Active Ingredient | Sodium Chloride (NaCl) |
| Preservative | 0.9% Benzyl Alcohol |
| Concentration | 0.9% (Isotonic) |
| Volume | 30 mL |
| Dosage Form | Injection, USP |
| Container Type | Multiple-Dose Vial (MDV) (Plastic Flip-Top) |
| Sterility | Sterile and Nonpyrogenic |
| Latex Status | Not Made with Natural Rubber Latex |
| Packaging / Sold As | Tray of 25 Vials per Carton |
| NDC Number | 00409-1966-07 |
| UNSPSC Code | 51191602 |
Primary Use
Used as a diluent or solvent to prepare or dissolve drugs for intravenous, intramuscular, or subcutaneous injection.